В журнале JOC - The Journal of Organic Chemistry (IF 3,6) опубликована статья, авторами которой являются сотрудники лаборатории азотистых соединений ( к.х.н., снс А.И. Таратайко, к.х.н., снс С.А. Добрынин, к.х.н, завлаб И.А. Кирилюк) и сотрудники лаборатории магнитной спектроскопии (д.ф.-.м.н., проф., гнс Е.Г. Багрянская, мнс С.Ю. Трахинина (аспирантка НИОХ, научный руководитель Е.Г. Багрянская), мнс Д.А. Кузнецов (аспирант НГУ, научный руководитель Е.Г. Багрянская)):
Sterically Shielded 3,3-Bis(hydroxymethyl) Pyrrolidine Nitroxides: Synthesis, EPR Spectra, Spin Relaxation and Reduction Rates
Sophia Yu. Trakhinina, Andrey I. Taratayko, Nargiz B. Asanbaeva, Danil A. Kuznetsov, Sergey A. Dobrynin, Elena G. Bagryanskaya, Igor A. Kirilyuk
J. Org. Chem. 2026, 91, 12, 4118-4129
Published on 17 March 2026
doi: 10.1021/acs.joc.5c02798
Abstract
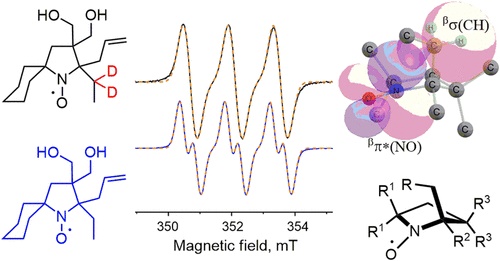
Sterically shielded nitroxides are known to be resistant to bioreduction. For this reason, they are attracting much interest as spin labels and probes for in cell and in vivo EPR and NMR applications. A series of sterically shielded 3,3-bis(hydroxymethyl)pyrrolidine-1-oxyls and 3,3-bis(hydroxymethyl)-1-azaspiro[4.5]decane-1-oxyls have been prepared via the formal [3 + 2] cycloaddition reaction of donor–acceptor cyclopropanes with propionitrile or isobutyronitrile, subsequent conversion of the resulting 1-pyrrolines to the corresponding N-oxides, and addition of allylmagnesium bromide or ethynylmagnesium bromide. Hydrogenation of multiple carbon–carbon bonds and oxidation were used to convert 2-ethynyl and 2-allyl nitroxides to 2-ethyl and 2-propyl. The EPR spectra of the new nitroxides are characterized by a large additional splitting with aH 0.23–0.42 mT, which was assigned to hyperfine coupling (hfc) with γ-hydrogen in the side chain based on the spectra of a partially deuterated derivative. In the spectra of the 2-ethynyl derivatives, the aH value was very sensitive to the polarity of the media. One of the nitroxides of this series, containing a spirocyclohexane moiety and an isopropyl group adjacent to the radical center, showed high resistance to reduction by ascorbate (k2 = 4.5 ± 0.1 × 10–3 M–1s–1). Some of these nitroxides demonstrate minor changes in phase memory time (Tm) upon heating from 80 to 120 K, similar to the dispirocyclic nitroxides.